|
Coulomb's LawStaff posted on November 10, 2006 |
;Coulomb's Law
| In 1785, Coulomb established the fundamental law of electric force between two stationary, charged particles. Experiments show that an electric force has the following properties: |
- The force is inversely proportional to the square of the separation, r, between the two particles and directed along the line joining the particles.
- The force proportional to the product of the charges q1 and q2 on the two particles.
- The force is attractive if the charges are of opposite sign and repulsive if the charges have the same sign.
|
| From these observations, we can express the magnitude of the electric force between the two charges as |
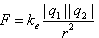 |
| Where ke is a constant called the Coulomb constant. In his experiments, Coulomb was able to show that the value of the exponent of r was 2 to within an uncertainty of a few percent. Modern experiments have shown that the exponent is 2 to a precision of a few parts in 109. |
The Coulomb constant has a value that depends on the choice of units. The unit of charge in SI units is the coulomb (C).
The coulomb is defined in the terms of a unit called the ampere (A), where current equals the rate of flow of charge. When the current in a wire is 1 A, the amount of charge that flows past a given point in the wire in 1s is 1 C. The Coulomb constant ke in SI units has the value |
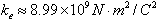 |
| The constant ke is also written |
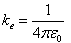 |
Where the constant 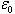 is known as permittivity of free space and has the value is known as permittivity of free space and has the value |
Where the constant 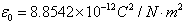 is known as permittivity of free space and has the value is known as permittivity of free space and has the value |
The smallest unit of charge known in nature is the charge on an electron or proton. The charge of an electron or proton has a magnitude
|
|e| = 1.60219 x 10-19C
|
| Charge and Mass of the Electron, Proton, and Neutron |
|
Particle
|
Charge (C)
|
Mass (kg)
|
|
Electron (e)
|
- 1.60219 x 10-19
|
|
|
Proton (p)
|
|
|
|
Neutron (n)
|
0
|
|